After the product patent had expired, the brand name pharmaceutical company sued generic drug manufacturers based on a manufacturing process patent. The Grand Panel of the IP High Court affirmed infringement under the doctrine of equivalents (DOE) for the medicinal compound's manufacturing process patent.
Summary of the case
The appellee, Chugai Pharmaceutical, jointly owns a patent for a manufacturing process of compounds including maxacalcitol. Chugai manufactures and sells Oxarol ointment, a therapeutic agent of keratosis, whose active ingredient is maxacalcitol. Chugai had owned a product patent regarding maxacalcitol. This patent expired on December 26 2010 after the patent term was extended.
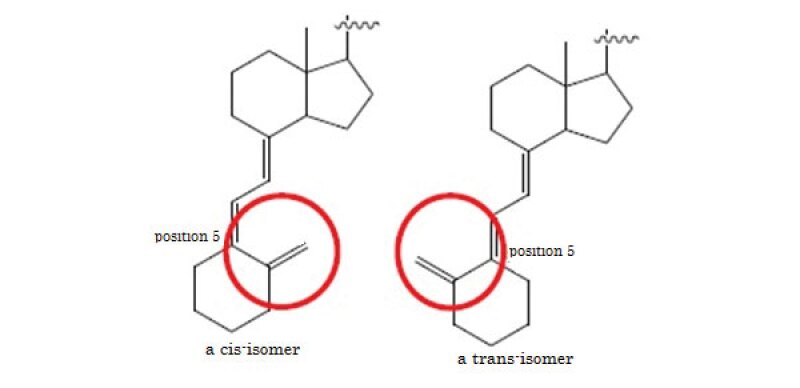
The appellants Iwaki Seiyaku etc sold generic Oxarol ointments and the appellant DKSH Japan had imported the active ingredients of the generic product from Cerbios Pharma and sold it. Chugai sought an injunction against the appellants' importation and assignment claiming that the appellants' manufacturing process of maxacalcitol preparations and their API was equivalent to the patented process. The appellants' process does not literally satisfy the claim elements because the structure of a starting material and an intermediate of the patented process is cis-isomer whereas that of the appellants' process is trans-isomer. The main issue was DOE.
Judgment of December 24 2014, Tokyo District Court
The Tokyo District Court (Presiding Judge Shimasue) affirmed satisfaction of five requirements of the ball spline case (judgment of February 24 1998, Supreme Court) and held that the appellants' process is equivalent to the Chugai invention. In February 2015, the Court granted a preliminary injunction.
Judgment of March 25 2016, The Grand Panel of the IP High Court
The Grand Panel of the IP High Court (Presiding Judge Shitara) affirmed infringement under DOE as follows:
(1) First requirement of DOE
Taking into account that the substantial value of a patented invention is defined depending on the degree of contribution in comparison with prior art in the relevant technical field, the essential part of a patented invention should be found based on the statements in the claims and the description, in particular, through comparison with prior art stated in the description.
If the degree of contribution of the patented invention is considered to be more than that of prior art, the patented invention is found as a generic concept in relation to part of the statements in the scope of claims (the corrected invention is an example of such a case).
If the degree of contribution of the patented invention is evaluated as not much more than prior art, the patented invention is found to have almost the same meaning as stated in the scope of claims.
However, if the statement of the problem, which is described as one that prior art could not solve, in the description is objectively insufficient in light of prior art as of the filing date (or the priority date), a characteristic part which constitutes a unique technical idea of the patented invention that is not seen in prior art should be found also in consideration of prior art that is not stated in the description. In such cases, the essential part of the patented invention is closer to the statements in the scope of claims compared to the cases where it is found only based on the statements in the scope of claims and the description, and the scope of application of the DOE is considered to be narrower.
The appellant's process is considered to have the characteristic part which constitutes a unique technical idea that is not seen in prior art in the statements in the scope of claims of the corrected invention. On the other hand, in the appellant's process, the point that a vitamin D structure that corresponds to "Z" of the starting material and the intermediate is not a cis form but a trans form, which is a difference from the corrected invention, is not the essential part of the corrected invention.
Therefore, the appellants' process satisfies the first requirement of DOE.
(5) Fifth requirement of DOE
Even if there is another structure that is outside the scope of claims, which a person ordinarily skilled in the art can easily conceive of as of the filing date as one that is substantially identical with the structure stated in the scope of claims and the applicant could thus have also easily conceived of another structure as of the filing date, this fact alone cannot serve as a reason for alleging that the applicant's failure to state that other structure in the scope of claims falls under the "special circumstances" in the fifth requirement of DOE.
However, if the applicant is objectively and externally regarded as having recognised another structure that is outside the scope of claims as a replacement for a different part in the structure stated in the scope of claims as of the filing date (for example, where the applicant can be considered to have stated the invention based on said other structure in the description or where the applicant stated the invention based on another structure that is outside the scope of claims in a paper, etc which he/she published as of the filing date), the applicant's failure to state said other structure in the scope of claims is considered to fall under the "special circumstances" in the fifth requirement.
Here, the applicant cannot be regarded as stating an invention using a starting material having a trans-form vitamin D structure in the corrected description, and it is not objectively and externally seen that the applicant recognised a trans-form vitamin D structure as a replacement for the starting material of the corrected invention as of the filing date. Therefore, the applicant's failure to state a structure wherein "Z" is a trans-form vitamin D structure in the scope of claims cannot be considered to fall under the "special circumstances" in the fifth requirement.
Therefore, the appellants' process satisfies the fifth requirement of DOE.
Practical tips
This judgment viewed the degree of contribution of the patented invention as important when finding the essential part of a patented invention. According to this criterion, the pioneering invention will enjoy wider scope of DOE.
The judgment held that prosecution history estoppel does not apply only by the fact that the applicant failed to state another structure in the scope of claims, but it applies if the applicant is objectively and externally regarded as having recognised another structure that is outside the scope of claims as a replacement for a different part in the structure stated in the scope of claims. According to Professor Tamura, this judgment acknowledged the doctrine of dedication.
This judgment has been appealed to the Supreme Court. The Supreme Court judgment should be watched closely.

|
Takanori Abe |
ABE & PartnersMatsushita IMP Building1-3-7, Shiromi, Chuo-ku, Osaka, 540-0001, JapanTel: +81 6 6949 1496Fax: +81 6 6949 1487abe@abe-law.comwww.abe-law.com










