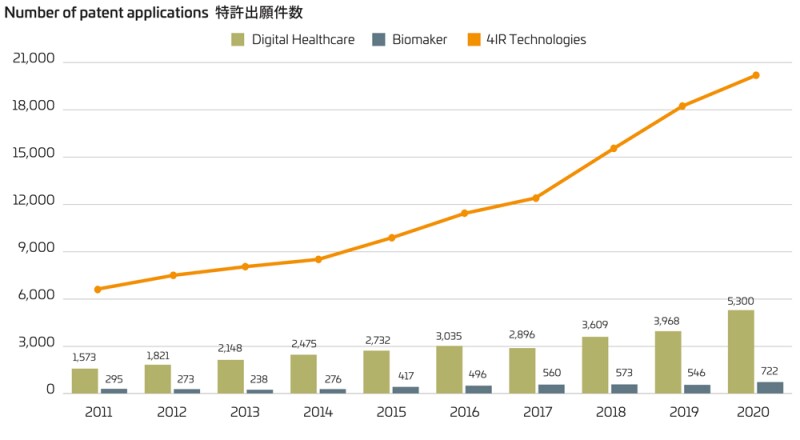
With the growing interest in Fourth Industrial Revolution (4IR) technologies, the pace of increase in relevant applications has been accelerating. The related technologies, as categorised by the Korean Intellectual Property Office (KIPO), cover eight technical fields: artificial intelligence (AI), big data (BD), internet of things (IoT), biomarker (BM), digital healthcare (DH), intelligent robot (IR), autonomous driving (AV) and 3D printing (3DP).
According to statistics released by the KIPO, the number of patent applications relating to the 4IR technologies in 2020 showed a sharp increase over 2019, despite the disruptive impact of COVID-19. In particular, patent applications in the field of digital healthcare and biomarkers increased more than 30% over the past year, in contrast to a 3.3% increase in the total number of patent applications in Korea.
In response to the convergence of cutting-edge technologies and the surge of 4IR technology applications, the KIPO underwent restructuring and established the Convergence Technology Examination Bureau to better examine the relevant patent applications. Further, the KIPO revamped its regulations for examining patent applications directed to 4IR technologies and published revised Patent Examination Guidelines for five key areas, including AI, IoT, and biotechnology.
Exemplary cases illustrated in the guidelines
In particular, the revised guidelines offer several illustrative, hypothetical cases to help resolve ambiguities in the standards for determining patentability of inventions in the field of digital healthcare, such as AI-based new drug development, AI-based diagnostic methods, and biomarkers, as summarised below.
Case one: new drug candidates using computer programs
One of the exemplary cases given in the new guidelines pertains to the patentability of an invention for developing a new drug by using a computer program, as defined in the following claims:
Claim 1. A method of discovering a drug candidate, the method comprising the steps of:
a) determining, by a computer device, a disorder-to-order transition region of a target protein using bioinformatics;
b) selecting, by the computer device, first candidate compounds capable of binding to the disorder-to-order transition region from among a compound library by performing molecular docking on the disorder-to-order transition region in conjunction with a library of specific compounds; and
c) selecting, by the computer device, a second candidate compound from among the first candidate compounds by performing a molecular dynamics simulation for the first candidate compounds and the disorder-to-order transition region.
Claim 2. A compound discovered by the method of claim 1, which is selected from the compounds of Formulas 1 to 3 (Formulas omitted herein).
Claim 3. A pharmaceutical composition for inhibiting metastasis of stomach cancer, comprising the compound of claim 2.
In the above hypothetical case, the inventive step would not be recognised if the screening method of new drug candidates uses a known computer program or algorithm to search for the compound and predict the actions thereof.
In addition, if the specification merely discloses the interaction of a new drug candidate with a target protein and the treatment effect for specific diseases based on an in silico method without confirming same with concrete experiments, it would not be considered to satisfy the utility or description requirement of the invention. As such, even when searching for new drug candidates on a computer, it would be important to provide, in the specification, experiments or data to corroborate that the new substance actually binds to the target protein and exhibits the treatment effect.
Case two: digital diagnostic method
Claim 1. A method of predicting the susceptibility of a subject to gastric cancer performed on a computer, comprising the steps of:
a) entering data of one or more gastric cancer allele mutations present in the subject into the computer;
b) comparing said data with a computer database comprising gastric cancer allele mutations and the gastric cancer information associated with the mutations; and
c) producing an indication for determining whether the subject is susceptible to gastric cancer, based on the comparison.
In Korean patent practice, a method for treating or diagnosing a human being is unpatentable for lack of industrial applicability. However, if a diagnostic method neither involves a clinical decision nor includes any step directly involving or affecting the human body, such a method would be deemed industrially applicable. As such, a method with steps relating to information processing technologies performed on a computer would be eligible for a patent.
The problem of patent eligibility may also be solved by claiming the diagnostic method as ‘a method of providing information’. Diagnostic methods exemplifying such claims in the Patent Examination Guidelines include “a method of predicting a cancer or providing information for cancer prediction by conducting AI algorithms on medical devices” and “a method of providing information for cancer diagnosis using an x-ray diagnostic device, comprising the steps of removing noises from x-ray images by a pre-treatment module and extracting the information for cancer diagnosis from the x-ray images by an AI module.”
Case three: biomarkers
Claim 1. A biomarker for predicting symptoms of Kawasaki disease, comprising polynucleotides which contain at least one polymorphism selected from the group consisting of rs2290692, rs28493229 and rs10420685.
Claim 2. A composition for predicting symptoms of Kawasaki disease, comprising polynucleotides which contain a polymorphism consisting of rs2290692, rs28493229 or rs10420685, or a combination thereof.
A claim reciting a biomarker limited by its medicinal use would be interpreted as claiming the substance itself, regardless of its use. In this regard, a claim for a biomarker is recommended to be written in the form of a composition claim or a kit claim, while being careful not to violate the unity of invention.
Taking the above case, as exemplified in the guidelines, if there is no common sequence or structural similarity among the polynucleotides, analysing the polynucleotides to detect certain diseases would not be regarded as a common technical feature that can be considered as an improvement over the prior art. Therefore, in order to satisfy the unity requirement, it would be desirable not to define the biomarkers in the form of a Markush group, but to list them individually, as shown in claim 2 of the above case.
Examination system for digital healthcare
In addition to the revision of the Patent Examination Guidelines, the KIPO made the following changes to support and facilitate the examination of digital healthcare applications:
Applications for personalised medicines and innovative new drugs are subject to expedited examination. Such expedited examination would secure a patent right in about five and a half months, which is more than 10 months shorter than the average period for regular examination.
As for convergence technologies, a joint examination would be conducted by a panel of three examiners, representing each sector. Since the establishment of the Convergence Technology Examination Bureau in November 2019 until April of 2021, 16.8% of the total cases examined went through the joint examination.
Click here to read this article in Japanese
Click here to read all the chapters from the Japanese Buyers' Guide 2021

Se Jeong Son
Partner, FirstLaw PC
T: +82 2 589 0010
Se Jeong Son is a Korean patent attorney and partner at FirstLaw. She has actively been involved in patent prosecution, IP trials and litigations and opinion work in the fields of pharmaceuticals, biotechnology and chemicals.
Se Jeong has worked on several landmark patent cases including invalidation trials and scope confirmation trials on behalf of multinational or domestic pharmaceutical companies. She has also successfully handled works regarding patent-approval linkage. As a leader of a team of professionals who primarily handle the cases of the firm’s Japanese bio/pharma clients, she has given a lot of lectures in Japan to share her expertise in the relevant fields. Having established close and trusted working relationships, she provides customised services for Japanese clients.
Se Jeong graduated from Seoul National University, college of pharmacy and received a LLM degree from University of Southern California, Gould School of Law.












